My thanks to Tony Thomas for this entertaining piece, first published at Quadrant Online
How scary is “ocean acidification”? Very scary. The previously scary “global warming” stopped 19 years ago, but do stay scared because all that CO2 since 1997 has instead been “acidifying” the oceans. Please imagine baby oysters dissolving in the equivalent of battery acid, and hermit crabs raising a nervous feeler to discover that their protective shells have disappeared. Curse you, horrible human-caused CO2 emissions!
In one celebrated episode involving Climate Science™, a lone oyster farmer in Maine put his oysters into a bucket and then found that the bivalves at the bottom were crunched because their shells were weakened.[1] Can any reasonable person ask for better scientific proof of ocean “acidification”?
“Ocean Acidification”, the evil twin of global warming, is scary because the chemistry is so simple. For example, the Australian Academy of Science in its curriculum for secondary schools, organizes an experiment for 16-year-olds where crushed ocean shells go into a test tube of sea water. You add acid or vinegar or something, and then watch the shells fizz and dissolve!
Two years ago, I noticed in Melbourne’s Fed Square a $50,000 competition for schoolkids for the best drawing about ocean “acidification”, sponsored by the green Ocean Ark Group. The theme was “Imagine losing all this color and life”. Guidance text included,
There are approximately 10,000 Coral Reefs and we are destroying one every other day…Left unchecked Ocean Acidification could trigger a Great Mass Extinction Event…
Now that union corruption has been exposed, maybe our next Royal Commission should be into Abuse of Children’s Intelligence, and the Academy and Ocean Ark could justify their teachings under cross-examination.[2]
Meanwhile, a trans-Atlantic team of top “ocean acidification scientists” has published a scary op-ed in the New York Times. Congrats to skeptic blogger Steve Milloy at Junkscience.com for successfully obtaining under FOI the emails among them collaborating over the op-ed draft. This material runs to 440 admittedly repetitious pages.[3] The named authors were Richard W. Spinrad, chief scientist of the U.S. National Oceanic and Atmospheric Administration (NOAA), and Ian Boyd, chief scientific adviser to UK’s Department of Environment.
The trove of FOI emails include some beauties. Here’s what NOAA’s Dr Shallin Busch had to say, privately, to her NOAA colleague Madelyn Applebaum on September 30 about the draft. They had been asked by the New York Times to sex it up with some specific hurts allegedly being caused by all this acidification. The editor asked,
It’s very interesting, but in order to work for us it needs to be geared more toward the general reader. Can the authors give us more specific, descriptive images about how acidification has already affected the oceans? Is the situation akin to the acid rain phenomenon that hit North America? What can be done to counteract the problem?
Dr Busch, who works for NOAA’s Ocean Acidification Program and Northwest Fisheries Science Center at Seattle, responded to Ms Applebaum:
Unfortunately, I can’t provide this information to you because it doesn’t exist. As I said in my last email, currently there are NO areas of the world that are severely degraded because of OA or even areas that we know are definitely affected by OA right now. If you want to use this type of language, you could write about the CO2 vent sites in Italy or Polynesia as examples of things to come. Sorry that I can’t be more helpful on this!
Dr Busch had the integrity to admit that science can cite “NO” significant ocean “acidification” impacts. But she was nonetheless happy for the article to include, as agitprop, the effects of natural CO2 venting through the ocean floor, as though this somehow corroborated the “acidification” story.
Dr Busch, in the course of vetting many drafts, also wrote to Applebaum:
Thanks for letting me chime in on this piece. My two general impressions are the following:
1) This article is mostly gloom and doom, which research has shown that people don’t respond to well. In fact, people just stop reading gloom and doom environmental stories. It could be good to highlight ways we can and are dealing with OA [Ocean Acidification] now and that we have an opportunity to prevent the major predicted impacts of OA by stopping carbon emissions before larger chemistry changes happen…
2) I think it is really important to resist the NYT editor’s impulse to say that OA is wreaking all sorts of havoc RIGHT NOW, because for ecological systems, we don’t yet have the evidence to say that. OA is a problem today because it is changing ocean chemistry so quickly. The vast majority of the biological impacts of OA will only occur under projected future chemistry conditions. Also, the study of the biological impacts of OA is so young that we don’t have any data sets that show a direct effect of OA on population health or trajectory. Best, Shallin. [My bolding].[4]
And here’s Dr Busch on the Great Barrier Reef. The “Chris” she refers to is Chris Sabine, director of the NOAA Pacific Marine Environmental Laboratory.
I’m not sure that I agree with Chris’s statement about the impact of OA on the Great Barrier Reef, [namely] ‘but underlying all of those factors is the fact that the corals are so stressed from ocean acidification that they can’t recover from those other impacts the way they used to be able to recover.’ Given my knowledge of the literature, OA is more of a future problem than a problem right now for the Great Barrier Reef. I think it is important to resist the NYT editor’s impulse to say that OA is wreaking all sorts of havoc RIGHT NOW, because for ecological systems, we don’t yet have the evidence to say that.
If you’ll permit a digression, Dr Sabine’s CV notes that not only was he a NOAA Employee of the Month in 2007 but his awards include:
Nobel Peace Prize (co-shared with Al Gore and other members of IPCC) – 2007
As an aside, I keep reminding these people of an IPCC ruling banning them from claiming Nobel Peace Prize status. Sometimes people like Deakin University’s Vice-Chancellor Jane den Hollander even referred to the (subsequently) abruptly-resigned ex-IPCC head, accused sex criminal and all-around dirty old man Rajendra Pachauri as a “Nobel Prize” winner.[5] Our own CSIRO is full of bogus Nobel Peace Prize winners. You can follow the preenings here, here and here, just for starters. The best solution would be for the IPCC to issue them all with lab coats recognizing their special status, e.g. with a purple satin sash, gold buckles and ostrich plumes.
Digression over, in the NYT’s other wordage and pics, we learn from the top scientists that sea butterflies, a food for salmon and herring, undergo shell weaknesses, “showing why ocean acidification is often called [by whom?] ‘osteoporosis of the sea’”. The NYT editor had been badgering the authors for pics to go with the article in order to spruik all this damage from “acidification”. The authors, via the indefatigable Madelyn Applebaum, were desperate to find such before-and-afters — a dauntingly hard quest, given there isn’t any damage and maybe never.
So what did Applebaum come up with to make the NYT happy? Why, twin pics from NOAA of sea butterflies (Pteropods) — the first had lived in a laboratory tub with “normal waters” (whatever “normal” means) for six days, and was in the pink of health. The other specimen inhabited a tub with “acidified water” for the six days. (Did the animal anti-cruelty people sign off on sea-butterfly torture?) Would you believe, the poor little acid-dunked Pteropod showed a tracery of white lines where the acid had etched its shell surface. What more pictorial proof of the harm of ocean “acidification” could a NYT editor (or climate scientist, or Academician) possibly require?[6]
All such articles have to point to harm to people too. We read emails that “Human health, too, is a major concern.” This is because the NOAA labs show toxic growths when water in the tubs is artificially acidified. So assuming (heroically) the same thing happens in the wild, the authors warn that people could get sick from eating acidy shellfish. Eating this nasty sea-stuff could “sicken, even kill, fish and marine mammals such as sea lions.” I imagine that right now, hundreds of NOAA IT people are modeling ocean-life die-offs based on that algae in a NOAA lab tub.
To climate scientists, the most important sea creatures in the entire planetary eco-system are not whales[7] or other coelecanths but Walt Disney’s celebrated clownfish. That’s because little Nemo is a sure-fire tear-jerker in any climate-catastrophe scenario.
In the long piece about the global oceans by the top dogs of US/UK climate science, we learn:
We cannot yet predict exactly how ocean acidification will affect connections among the world’s many different marine organisms, but we do know the consequences will be profound. [i.e. we don’t know but we do know. Send more grant funding immediately.] Research already points to the unnatural behavior of coral clownfish in an acidified environment. These fish wander farther from their natural protection, making them more vulnerable to predators.
This published reference to clownfish was the fruit of much angst involving the UK department’s determination to shoe-horn Nemo into the PR exercise.
Jane Phenton, Senior Flack for UK Environment Dept, 30/9/15:
The [UK] team have added some examples (Nemo the clown fish a particularly good one I think!) and a few thoughts.
Comments on a draft:
Hearing loss/impairment in Nemo, the coral clownfish, is just one of many potential impacts that have been identified in laboratory studies…
Oh no! Nemo, too, has been subjected to laboratory acid torture, but in a good cause. The boffins found “he” began wandering further from his protective home, inviting danger. But couldn’t Nemo, if still right side up in the tub, now be fitted with a sea-going Cochlear implant? (editor: shouldn’t that be a conchlear implant?)
However, things got more complex, because, says a NOAA scientist,
Apparently one study called Nemo ‘deaf’ – problem was attributed to brain damage that affected capability to hear. I’ll word carefully.
Someone else chips in,
He [Nemo] can’t smell his predators when they are near, and engages in risky behavior, making him more vulnerable to predators.
Someone else bells the cat, or fish, by pointing out that Nemo’s lab tub isn’t literally acidic, just less alkaline. And, anyway, Nemo might work out how to adapt [assuming Nemo’s brain damage isn’t terminal].
Then on September 30 a NOAA heavy Dr Chris Sabine, Director, Pacific Marine Environmental Laboratory, weighs in to Madelyn:
I have asked everyone I can reach and nobody is aware of a study that suggests that Nemo’s hearing would be impaired by ocean acidification. I did find one article on the web that suggested the opposite. I am aware of studies indicating that Nemo would lose sense of smell or ability to detect predators and therefore would be more likely to be eaten. Perhaps you can ask the UK people to check on that sentence. Chris [my emphasis]
Be aware that these collaborators obsessing about Nemo are taxpayer-funded scientists and PR flacks, all hard at work on a journalism piece to puff their organisations.The article continues,
In the past three decades, the number of living corals covering the Great Barrier Reef has been cut in half, reducing critical habitat for fish and the resilience of the entire reef system.
I looked up the 2014 Great Barrier Reef Marine Park Authority’s Outlook Report which re-quoted a 2009 study, (p31),
There is little detailed information about the status and trends of many habitat types within the Great Barrier Reef … However, there is some evidence of a small decline in coral reef habitat over recent decades.
A “small decline”? So what’s this halving that NOAA is talking about? It seems to come from a 2012 paper by De-ath et al from the Australian Institute of Marine Science.
By analyzing 27 years of data, the authors found that the Barrier Reef’s coral cover was down from 28% to 13.8% by area, in other words, half the initial coral cover had been lost. But why? The losses were due to cyclones (48%), crown-of-thorns starfish (42%) and coral bleaching (10%) – none of this involves the “acidification” peril. And the pristine northern Reef area showed no decline. If it wasn’t for the cyclones, starfish and bleaching, the coral overall would have grown by nearly 3% a year. Even with cyclones and bleaching, the coral would grow by nearly 1% a year if the starfish were neutralized. To stay politically correct, the authors added that climate change had to be (somehow) stabilized, otherwise there would’s be more bleaching and cyclones, they think.
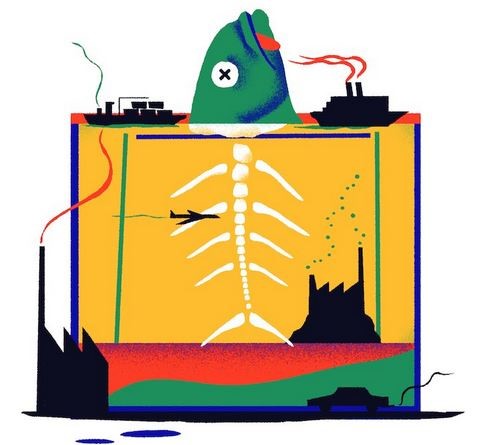
The NYT International piece was published under the ludicrous headline: Our Deadened, Carbon-Soaked Seas. A graphic (below) showed a big fish whose head above water was intact but whose body underwater was reduced to an acid-etched skeleton.
The scientists’ preferred headline had merely been “In a high CO2 world, dangerous waters ahead” and the authors had nothing to do with the lurid graphic. But the NYT likes to greenwash its readers with terrifying climate capers. The broadsheet gives several pages of boilerplate specs for contributors on fact-checking, and has an army of its own fact-checkers to ensure against embarrassing corrections. But this “professionalism” is trumped by a headline-writer’s whim and an artist’s ignorant sensationalism. As with our own demented and dying Fairfax papers, sensationalism is given the tick of shock-horror approval all the way up the editorial chain.
The top-tier science authors commiserated among themselves about the “quite inflammatory” re-write of their headline, but made not one word of complaint to the NYT. After all, the luridness was in the right direction – mega-scariness – and the authors were keen to get more NYT coverage in future. Principles be damned.
The NYT article made a lot of mileage out of US west-coast oyster industry problems ostensibly caused by ocean currents pushing “acidified” water towards the oyster beds, causing “baby oysters” to expire. The infant oysters had in fact been killed by a faecal organism Vibrio tubiashii from sewage.) And in any event, that pesky Dr Busch throws in an email saying
In fact, production in the Washington oyster industry is higher now than at the start of the [supposed acidification] crisis…Just as an FYI, we can’t yet attribute any large patterns in shellfish yield to OA [ocean acidification].
Dr Busch also wrote, re specific fish communities, “It might be good to mention that some species will be harmed by OA, some will benefit, and some won’t respond at all!” This is complete heresy, as global warming must always be presented as a bad thing. But Dr Busch knew what the NOAA playbook demanded and constructed a new draft paragraph dotted with the conditional — words like “may affect some fish populations” and “may” reorder ecosystems. In this way any references to positive impacts on marine life are made to disappear.
The Australian Academy of Science in its educational materials is likewise unable to actually admit that lower pH can have positive impacts on sea life. Instead, it glooms,
not all calcifying animals react in the same way to lower pH conditions. But although some animals and plants may not fare so badly as others, the impacts upon marine biodiversity have the potential to be severe.
The NYT piece revels in ridiculous analogies and apparently-massive numbers isolated from any planetary context, eg:
Over the past 200 years, the world’s seas have absorbed more than 150 billion metric tons of carbon from human activities. Currently, that’s a worldwide average of 15 pounds per person a week, enough to fill a coal train long enough to encircle the equator 13 times every year.
Hence
ocean and coastal waters around the world are beginning to tell a disturbing story”. (My emphasis. Note how the bolded words fudge that there is actually no story so far).
Innocent readers might imagine NOAA embarked on this new op-ed project to educate the public about a serious scientific matter. No, the project’s prime and explicit function was to puff NOAA as a funding-worthy institution, and to add ammunition to the COP21 climate talks in Paris in December, 2015. As Dr Libby Jewett, director of NOAA’s Ocean Acidification Program, briefed:
The article would fit well with NOAA’s resilience and observational priorities and could go an important distance in recognizing NOAA’s leadership in growing an international ocean observing system…We want visibility for NOAA’s pioneering global leadership to be prominent, too!
The final 250 words — 20% of the 1250-word NYT piece — are all about the need to send money to the scientists for their planet-saving endeavours,
Smart investments in monitoring and observing are critical to building resilience and hedging risks that can directly affect economies at all levels. There is urgency to such investments. The U.S. National Oceanic and Atmospheric Administration conducts round-the-clock monitoring of global CO2. The rate of increase has never been higher than during the past three years, accelerating the ocean acidification process… We ignore the risks of ocean acidification at our own peril, and that of future generations.
Tim Flannery, head of Australia’s Climate Council, is of the view that CO2 falling into the ocean produces “carbolic acid” or phenol, that useful disinfectant which can still be bought on eBay in the form of soap bars. Flannery is, as always, correct in terms of the prevailing hysteria, if not real-world facts. His prophecy is affirmed by Ocean Acidification International Coordination Centre (OAICA) and the International Atomic Energy Agency (IAEA), which agree that
Too much carbon is flooding the ocean with carbolic acid, with devestating (sic) effects on life in the sea.
This is devestating (sic) news for chemistry textbooks.[8]
Here’s a contrary view to all that. The Center for the Study of Carbon Dioxide and Global Change, run by sceptic scientists, agrees with the orthodox group that, since pre-industrial times, the oceans have become less alkaline by about 0.1 pH unit. But it considers results from modeling that posits a further pH reduction of between 0.3 units to 0.7 units by 2300 to be far-fetched. It marshaled about 1100 peer-reviewed studies on impacts of lower pH on ocean life and, after excluding those with wildly unrealistic assumptions, checked the rest in terms of five factors: calcification, metabolism, growth, fertility and survival. It plotted the experimental results involving pH falls from 0.0 to 0.3, the latter number being what the IPCC predicts for 2100, and found that the fall in pH led to
an overall beneficial response of the totality of the five major life characteristics of marine sea life to ocean acidification, which result is vastly different from the negative results routinely predicted by the world’s climate alarmists.
It said the results would be even more positive if studies had also allowed for the ability of generations of sea life to adapt to changed conditions. The studies testing lower pH on life forms typically involved a mere four days duration and some trials lasted a mere few hours, preventing any favorable evolutions, it said.
Footnote: My studies in high-school chemistry ceased at age 16, but here’s my take on ocean acidification technicalities.
The oceans’ alkalinity (pH) varies from place to place, in a range 7.9 to 8.3 on a logarithmic scale where 14 is most alkaline (or basic), 7.0 is neutral and below 7 to zero is acidic. The log scale means each change of one unit is ten times the value of the adjacent unit.[9]
The scare term “ocean acidification”[10] first popped up in Nature in 2003, followed by the Royal Society in 2005[11], and has since been seized on as a substitute frightener, given that global warming has stalled. Climate scientists now “calculate” that the average ocean alkalinity has declined from 8.2 to 8.1 on the scale since pre-industrial times, except that the measurement error margin is several times the alleged reduction (and each of the five oceans has its own pH characteristics). pH levels at given points can also swing markedly even within the 24-hour cycle.
In past geological ages C02 levels in the atmosphere were ten or more times what they are now (400ppm) and ocean life thrived. Indeed our current fossil fuels are the residue of vast oceanic life that thrived and died in such super-high CO2 environments.
In the parts of the oceans where alkalinity is low (i.e. tending towards neutral), fish, corals, and sea flora have managed and adapted perfectly well. Freshwater lakes and rivers are slightly acidic (pH of 6 to 8), as is rainwater, pH 5.6, and drinking water, 6.5 to 7.5. Life has adapted and thrives in fresh water notwithstanding the, ahem, “acidification”.
Hat-tips to Dennis Ambler and John McLean for some assistance.
Tony Thomas blogs at No B-S Here, I Hope
[1] An earlier draft: In Maine, clam farmers can no longer fill their buckets to the top because shells on the bottom will shatter from the weight. The lone clam farmer later bred into multiple clam farmers all ostenisibly reporting the same bucket problem.
[2] “…Some of the octopuses in the ocean can’t breathe from gas pollution … I started off my poster design of drawing the octopus first and wanted to make it look as if he was dying. Then I drew dead coral coming and surrounding him like there’s no escape from the acid ocean…” – one child’s entry in the Victorian schools contest for “ocean acidification” art
[3] Strangely, NOAA has been fighting tooth and nail to thwart Republican congressmen’s requests for its emails about a NOAA study by Karl et al purporting to show there has been no 15-year pause in warming . It is now conceding defeat and starting to hand over documents.
[4] The 2014 5th IPCC report Summary for Policymakers, written by politico-bureaucrats, waxes fearful about ocean acidification. But the scientists themselves in their non-sexed-up findings in body text, WG11 Chapter 6, say, for example,
# “Few field observations conducted in the last decade demonstrate biotic responses attributable to anthropogenic ocean acidification” pg 4
# “Both acclimatization and adaptation will shift sensitivity thresholds but the capacity and limits of species to acclimatize or adapt remain largely unknown” Pg 23
# “To date, very few ecosystem-level changes in the field have been attributed to anthropogenic or local ocean acidification.” Pg 39
[5] “As well as helping fulfil Deakin’s prophecy, teaming up with TERI is a major coup for Deakin University. The organisation’s Director-General is Dr Rajendra K. Pachauri, the Nobel Prize winner”.
[6] Or the Australian Academy of Science, which has recycled the Pteropod before-and-after pics for schoolkids.
[7] Some say whales aren’t fish, but I say they look more like fish than coelecanths.
[8] The OAICC and IAEA warn that climate is putting the world “in a dangerous position, just as the US was when it was bombed at Pearl Harbor… This segment reports on “global warming’s evil twin”, ocean acifidication, which results from too much carbon in the water.” The audio segments begin with air-raid sirens and crashing bombs.
[9] Vinegar, for example, at 2.5, is almost a million times more acidic than seawater.
[10] Compare it with its twin verbal Orwellism, “carbon pollution”
[11] With authors in common. By linearly extrapolating 18 years data from a single Pacific Ocean Station Aloha, both parties forecast perilous ocean acidity by 2100 – and even took the perils out further to 2300. This became ‘settled science’.






Th white cliffs of Dover were laid down in the Cretaceous when the CO2 level was a minimum of 4 times the current level. Where was any adverse effect?
More chemistry orientated is that about 98% of CO2 dissolved in the oceans is there as gas. So if CO2 causes Global warming then less CO2 will dissolve.
Secondly the reaction to carbonic acid is pH dependent; it is strongly towards the carbonic reaction above a pH of 9.5 but if the oceans were that alkaline not much sea life would survive anyway. Conversely as the pH gets lower the reaction to carbonic gets slower. Around about pH 5.5 the “acidification” stops. So higher levels of CO2 will result in less and less effect.
Thirdly, the conversion to carbonic is affected by the level of calcium and magnesium in sea water, which precipitate it out as insoluble carbonates.
Fourthly, the actual reaction they are waffling about is to the bicarbonate ion. Sodium bicarbonate solution is still alkaline.
Fifthly, there is evidence from those tropical reefs with 100% CO2 bubbling into sea water under about 2 atmospheres pressure results in a pH of 6.2. For all that there is no evidence that that pH is effecting the coral around the gas bubbles, possibly because the pH rapidly jumps to alkaline within 20-30 centimetres as the calcium & magnesium salts buffer the minor acidification.
As a final point I note that the ‘experiments’ involved acids that had soluble calcium and magnesium salts and in the case of acetic acid escape any rational explanation for enough being released naturally to give the effect they claim.
This is a disgraceful scare campaign based on very little knowledge of chemistry, buttressed by “models” of previous extinction episodes e.g. the Permian extinctions, where high levels of CO2 are ASSUMED to be responsible, thereby circular ‘reasoning’ ‘proving’ CO2 caused ocean acidification. No one asks what gases the Siberian traps eruption actually released; were they hydrogen sulphide or sulphur dioxides? Both of these are far stronger acids than CO2 so might have caused “acidification”.
A great post! This is the first one I have read this year and what a hoot this one is. Thanks for posting it.
I mentioned this one at a couple of other places. Oddly, every time I mention this site or link to it my comment goes into deep moderation at wuwt. I suppose they can’t handle non-lukewarmer science. Oh well.
The science experiment for kids was priceless. I am going to ask a friend who teaches science if she uses that one. I’ll report back if I can get an answer.
~ Mark
The special lab coats for nobel prize winning climatologists made me laugh out loud. 🙂
The coats suitable for certain lukes would have leather straps and steel buckles rather than purple sashes and gold buckles though. Tar and feathers rather than ostrich plumes too. 😉
ARGGGHHHhhhhhh,
As a Chemist with a lot of geology courses, Ocean Acidification really makes me want to scream.
I can not find the link (old WUWT comment?), but the change in pH ” the oceans have become less alkaline by about 0.1 pH unit.” is because the ClimAstrologists tossed out a lot of the data. If all the data is included no change.
Number two. Check out Dr Segalstad’s work
Older essay: Questions have been raised about how strong this buffer is….
New work for the NIPCC Report
The other point over looked is where all the additional buffers are coming from.
The oceans are highly buffered by calcium Ca++ (and magnesium Mg++)
SEE: http://www.advancedaquarist.com/2002/3/chemistry
Calcium is the main cation in river water, followed by Na and Mg, then K. – All that dissolved and eroded stone washed down our streams and rivers into the sea.
Mods, please fish (snicker) out my last comment. (this is #3)
And the final slap upside the head with reality:
No one could keep a reef aquarium in the house, where it’s common for CO2 levels to be over 1000 ppm, if CO2 in the air was a real problem.
If WordUnimpressed ate comment #2 you can find it here https://stevengoddard.wordpress.com/2016/01/01/cold-january-march-is-weather-warm-december-is-climate/#comment-563010
(We are having lots of fun playing with the trolls at Tony’s)
Thanks for the quick action mods.
And a very Happy, prosperous and uneventful New Year to all of you.
Hi Gail,
Thanks for the informed comment, and a Happy New Year to you too.
Hi Tallbloke,
I was happy to toss a bit of information your way. Often I just read since much is over my head math wise. (The calculus is forty years old and unused)
This extract from a NASA feature in 2008 may be of interest here.
‘The Ocean’s Carbon Balance’
“When we started in the 70s and 80s, we had this concept: we’ll measure [carbon dioxide concentrations in the ocean] 10 years later, and we’ll just see the anthropogenic input,” says Feely. “We had very simplistic ideas that the anthropogenic changes would be the only changes we would see,” he adds a little ruefully.
Feely and his colleagues saw changes, but they weren’t at all the changes they expected. Carbon concentrations in the ocean did rise as atmospheric carbon dioxide skyrocketed, but in 2006, Feely and several colleagues announced that the equatorial Pacific seemed to be venting more carbon dioxide to the atmosphere between 1997 and 2004 than it had in previous years. And in 2007, Ute Schuster and Andrew Watson, oceanographers from the University of East Anglia, reported that the amount of carbon that the North Atlantic Ocean soaked up decreased by a factor of two between 1994 and 2005. The ocean, or parts of it, seemed to be taking up less, not more, carbon.” [bold added]
So the whole carbon cycle is much more complex than headline writers and shouty warmists would have us believe.
[…] as possible, and this truth was revealed recently in a series of emails behind the publication of an ocean acidification disaster story in the New York Times using this […]
[…] as possible, and this truth was revealed recently in a series of emails behind the publication of an ocean acidification disaster story in the New York Times using this […]